|
March 2011, Volume 33, No. 1
|
Update Articles
|
Assessment and management of diabetic albuminuriaMan-kuen Cheung 張文娟, Fai-ying Wong 黃輝瑩, Jun Liang 梁峻 HK Pract 2011;33:14-19 Summary Microalbuminuria is the earliest clinical evidence of nephropathy in diabetic patients. Early detection and intervention can help to slow or even revert the progression to more advanced stages of nephropathy. This article aims to review and discuss recent trends in assessing and management of diabetic albuminuria. In general, urine dipsticks should not be used to identify albuminuria. Albumin to creatinine ratio (ACR) is the preferred test, as it is a validated, more convenient and more accessible method. Patients with microalbuminuria, compared with those with macroalbuminuria, will have different implications in therapeutic interventions. Proteinuria in a diabetic patient may not necessarily be attributable to diabetes. 摘要 蛋微量蛋白尿是糖尿病腎病最早的臨床證據。早期診斷及治療有助於延緩甚至逆轉腎病向晚期發展。本文旨在對糖尿病蛋白尿的診斷與管理的最新趨勢進行綜述和討論。一般不採用尿液試紙來診斷蛋白尿,而應首選白蛋白-肌酐比(ACR),這種方法有效、方便且易行。與有大量白蛋白尿的病人相比,對微量蛋白尿的病人應有不同的治療方法。糖尿病人出現蛋白尿不一定由糖尿病引起。 Introduction Diabetic nephropathy is becoming the leading cause of end-stage renal disease wordwide.1-4 The appearance of microalbuminuria is the earliest clinical evidence of nephropathy.5 Previous studies showed that each year, 2% of patients without albuminuria progress to microalbuminuria, 3% with microalbuminuria progress to macroalbuminuria, and 2.5% with macroalbminuria progress to end stage renal failure.6 However, if detected early, such progressions can be slowed, arrested, or even reverted.5 In the latest Hong Kong Reference Framework for Diabetes Care for Adults in Primary Care Settings 2010, which was just released by the Food and Health Bureau, it was recommended that screening for microalbuminuria should be done annually in all Type 2 diabetic patients. This article discusses recent trends in assessing diabetic albuminuria, and reviews the current literature on the management of diabetic patients with albuminuria. Assessment tools for albuminuria in diabetes
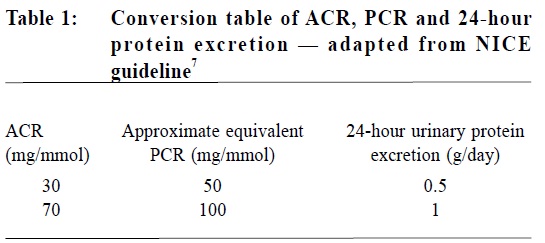
Urine dipsticks Detection of proteinuria at the point of care using dipsticks has been widely used as a screening test to guide further quantitative urine test. A recent analysis in a NICE guideline showed that reagent strips used to detect proteinuria in routine clinical practice are predominantly sensitive to albumin, not to total protein. No one type of reagent strip performed better than the others. The positive predictive values of reagent strips for detecting albuminuria ranged from 71-91%. The negative predictive value of reagent strips varied according to the cut-off values used to define albuminuria, ranging from 76-100%.7 An Australian paper noted that at a specificity of 67%, the sensitivity of dipsticks for proteinuria was 90%. The specificity of dipsticks was considered low, which would result in a high proportion of the population being recalled for more tests before being declared false positives.8 A UK study also showed that dipstick for proteinuria has an acceptable sensitivity but poor specificity.9 Therefore, reagent strips should not be used to identify proteinuria among diabetic patients unless they are capable of specifically measuring albumin at low concentrations and expressing the result as an ACR.7 Timed urine collection: 24-hour urine albumin measurement The establishment of proteinuria as the single most important clinical variable for predicting the risk of future chronic kidney disease progression and guiding the therapy in chronic kidney disease (CKD) has been based on evidence using 24-hour urine total protein measurement.10 Therefore, measurement of albumin excretion in a 24-hour urine collection has long been the "gold standard" for quantitative evaluation of albuminuria in diabetic patients. However, collection errors due to improper timing and missed samples may lead to significant over- and under-estimation of albuminuria. There is also obvious inconvenience to patients.11 Recently, spot urine collections (ACR or PCR) have been widely adopted as more convenient alternatives. Spot urine collections: The measurement of protein on a single-voided specimen of urine would be more convenient but is limited by the obvious problem of variable urine concentration resulting from variations in hydration status. To overcome this, it was proposed that protein measured on a single-voided urine specimen should be expressed as a ratio to urinary creatinine, a marker of urine concentration.10 Albumin-to-creatinine ratio (ACR) A recent study found that ACR accurately predicted both an abnormal 24-hour urine albumin ≥ 30mg / 24-hour and frank albuminuria at ≥ 300mg / 24-hour or ≥ 24-hour.12 Detection of early diabetic kidney damage at a stage when therapy could be usefully intensified is now nearly universally through urinary ACR.13 A first-morning sample is preferred because of the potentially higher correlation with 24-hour albumin excretion, but a random sample is considered acceptable if a first-morning specimen is not available.11 Protein-to-creatinine ratio (PCR) Diabetic or not, a highly significant linear regression was found between spot morning PCR and 24-hour urinary protein excretion rates.7 PCR accurately predicted both an abnormal 24-hour urine protein ≥ 150mg/ 24-hour and significant proteinuria above 300mg/ 24-hour.12 However, PCR becomes a less accurate predictor of 24-hour urinary protein excretion in the higher values.7 The approximate equivalents that allows conversion of ACR values to PCR or 24-hour urinary protein excretion rates is shown in Table 1.
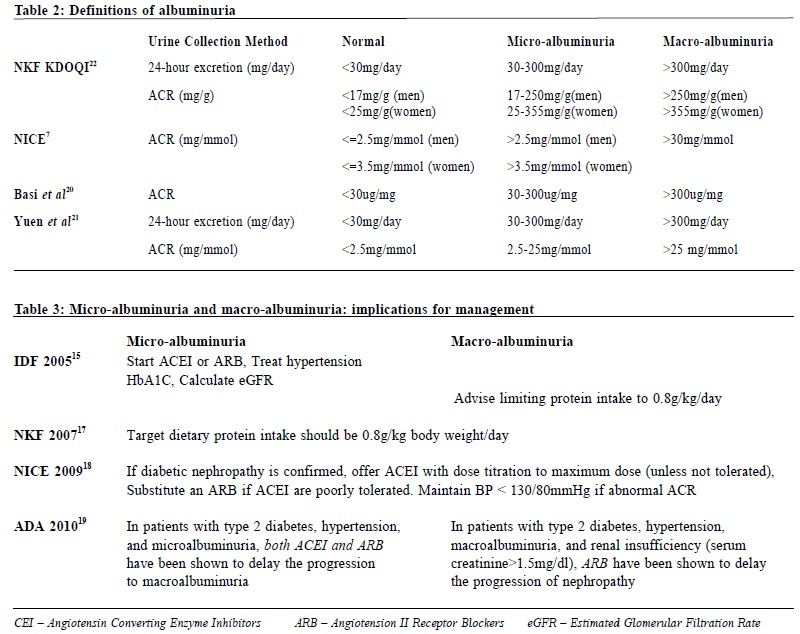
It is recommended that ACR is preferred for detection and identification of proteinuria, as it has greater sensitivity than PCR for low levels of proteinuria. PCR can be used as an alternative in quantifying and monitoring proteinuria. ACR is the recommended method for people with diabetes.7 Definition of albuminuria Definition of albuminuria varies according to the tests used, and different cut-off values are used to define micro- and macro-albuminuria in different clinical guidelines. (Table 2).
Algorithms for assessment of DM – what do guidelines say? The World Health Organization (WHO) in 2002 suggested measuring ACR in a first pass morning urine, and then confirming with overnight or 24-hour urine collection. Urine ACR > 200 microgram/min or > 300mg/ 24-hour were the cutoff point for DM nephropathy.14 International Diabetes Federation (IDF) in 2005 proposed to check urine with a dipstick. If dipstick test is positive, obtain a urine PCR after urinary tract infection is ruled out. If dipstick test is negative, obtain a urine ACR. If PCR or ACR is raised, repeating the test twice more over the following 4 months were suggested. Albuminuria or proteinuria is only confirmed if there is raised ACR or PCR finding on two of the three occasions. If both repeat tests were not raised, annual screening should be performed.15 In 2006, WHO recommended ACR as the preferred method of annual screening. Confirmation is required through repeated testing and elevated albumin excretion rate should be confirmed by repeated test.16 In 2007, National Kidney Foundation recommended to check urine ACR annually. If ACR is positive in the absence of urinary tract infection, it should be repeated for 2 more times within 3-6 months. Albuminuria is confirmed if ACR is positive in at least two of the three samples.17 The same line of thinking has been adopted by National Institute for Health and Clinical Excellence (NICE) in 2009 18 and American Diabetes Association in Standards of Medical Care in Diabetes 2010.19 In short, as more and more studies confirmed the close correlations between results from spot urine collection and timed urine collection, there is an international trend to adopt urine albumin-to-creatinine ratio (ACR) and urine total protein-to-creatinine ratio as validated, more convenient and more accessible alternatives for albuminuria assessment. It is important to note that, when interpreting ACR results, other causes of microalbuminuria, such as urinary tract infection, severe hyperglycaemia and cardiac failure, should be excluded. Vigorous physical activity, contamination with blood or other renal disease could potentially cause a false result – a phenomenon not too infrequently encountered in clinical practice. Micro- and macro-albuminuria – implications for management Diabetic albuminuria is divided into micro- and macro-albuminuria. Their implications for management are summarized in Table 3. Treatment with ACEI or ARB can be initiated irrespective of blood pressure level, unless there is significant hypotension (BP<100/60mmHg). The target for blood pressure control is <130/ 80mmHg. Combinations of ACEI and ARB have been shown to provide additional lowering of albuminuria.23 However, the long term effects of such combinations on renal and cardiovascular outcomes have not yet been evaluated in clinical trials and they are associated with increased risk of hyperkalaemia. Diabetic kidney disease One point to note is that in patients with diabetes mellitus (DM) and chronic kidney disease (CKD), the CKD may not necessarily be attributable to DM, as there may be other causes of CKD developing in a patient with DM. CKD should only be attributable to DM if:
1. macro-albuminuria is present, or
This is because in type 2 DM with microalbuminuria, only about 40% of renal biopsy results show diabetic changes typical of those seen in patients with type 1 diabetes.17 Non-diabetic causes of proteinuria should be considered in patients with active urinary sediments e.g. RBC or casts, and type 2 DM without DM retinopathy. In such circumstances, specialist's opinion should be sought. Conclusion The development of new diagnostic processes is a step forward in the advancement of medicine, with improvement in accessibility, convenience and thus adherence. By understanding the development of the new assessment tools for albuminuria in diabetic patients, and getting familiar with the prevailing international guidelines, clinicians are better equipped to design an appropriate complication screening programme for their diabetic patients. Depending on the different levels of diabetic albuminuria, appropriate interventions can be provided, including management of modifiable risk factors (especially dysplipidaemia), monitoring and review of lifestyle issues on a regular basis, and appropriate pharmacological treatment.
Man-kuen Cheung, FHKAM (Family Medicine), FHKCFP
Associate Consultant, Fai-ying Wong, FHKCFP, FRACGP Resident, Jun Liang, FHKAM (Family Medicine), FHKCFP Consultant, Department of Family Medicine, New Territory West Cluster, Hospital Authority. Correspondence to : Dr Man-kuen Cheung, Department of Family Medicine, Tuen Mun Hospital. Email: cheungmk1@ha.org.hk
References
|
|